The Shield™ blood test for
Colorectal Cancer (CRC) screening:
Trusted by Doctors.
Powered by Pioneers.
Order Shield
in Quanum Lab Services Manager or your EHR
or enter test code 18441
Shield is performed and billed through Guardant Health
*Patrick Dempsey is a paid partner of Guardant Health.
*Patrick Dempsey is a paid partner of Guardant Health.
Why Shield?
Guideline
Included
Recommended by the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for CRC Screening1*
See recommendations
Proven
Performance
90%+ adherence2† and 2x+ screening increase3,4‡
100,000+ Shield blood tests have been completed5
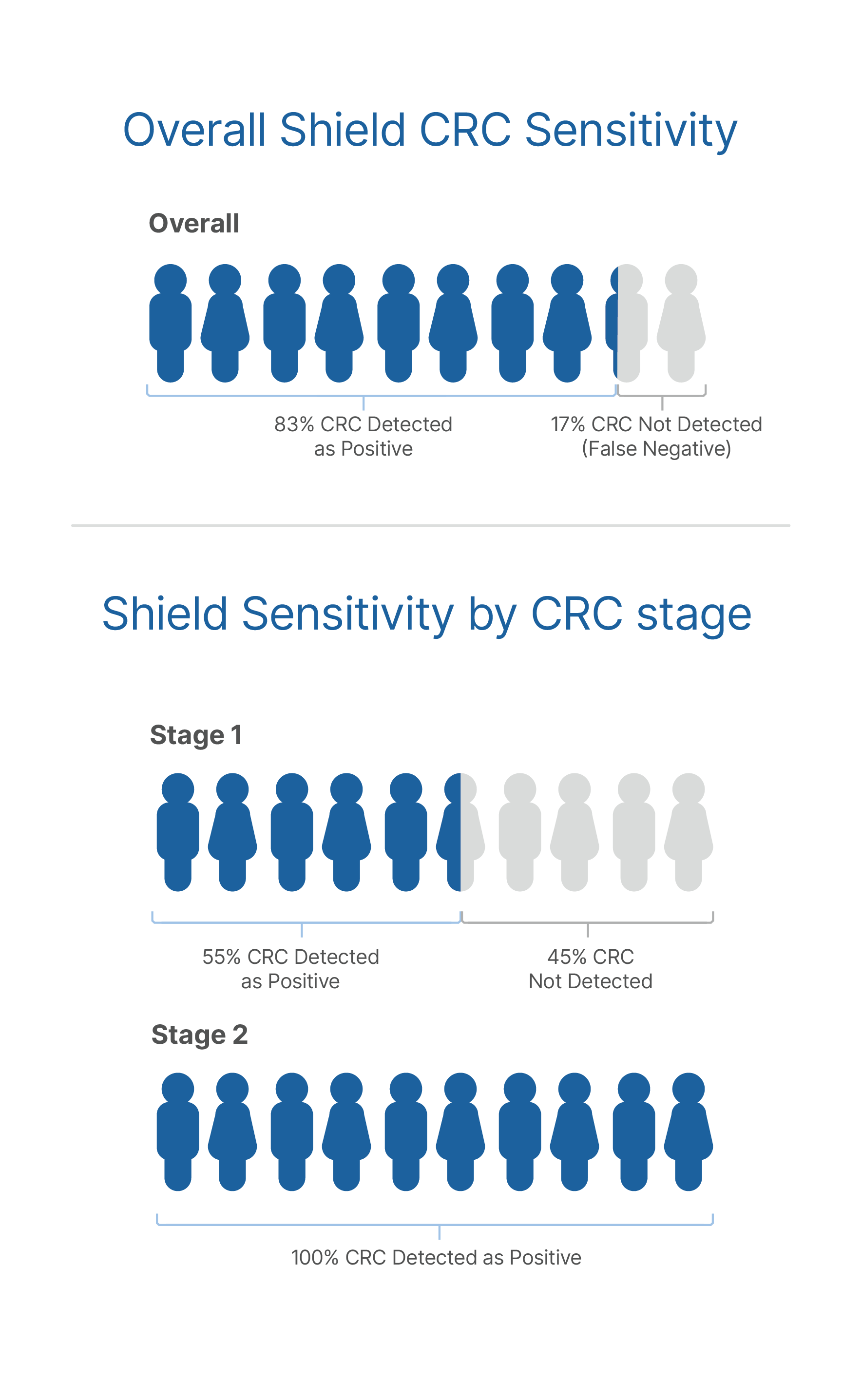
Accuracy published in the NEJM6
View performance data
Powered by
Guardant Health
Shield is backed by Guardant's 10+ years of experience and 1+ million blood tests performed5
See what powers ShieldNEJM=New England Journal of Medicine.
*NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
†Data from real-world clinical settings between May 2022 and June 2024 based on Laboratory Developed Test (LDT) usage of Shield, prior to FDA approval.
‡Based on 2 studies. Study 1 compared screening completion with ‶usual care″ (fecal immunochemical test [FIT] and colonoscopy), and an ‶intervention″ (usual care + Shield) in over 1000 patients, age 45-75 years, who initially were offered but did not complete FIT in the previous 3 to 9 months at a large, integrated health system. Study 2 compared adherence to standard of care vs Shield use in <250 patients age 45-84 years at 2 primary care clinics affiliated with a large, not-for-profit integrated regional health system. All data based on LDT usage of Shield in 2022 and 2023 (Study 1) and 2024 (Study 2), prior to FDA approval.
Hear from healthcare professionals using Shield to help more patients get screened

“Shield has been a game changer in getting people screened that have various barriers in their path.”
Tom Wieme
Physician Assistant - Certified
Tom is a paid spokesperson of Guardant Health.
Shield™ is the only FDA-approved blood test for CRC screening with Medicare coverage*
out-of-pocket cost for eligible Medicare Part B patients7
<$50 out-of-pocket cost for a majority of insured patients, including those who qualify for financial assistance
<$50 out-of-pocket cost for a majority of insured patients, including those who qualify for financial assistance
CMS covers the cost of a follow-up colonoscopy after a positive Shield test result
Shield is also covered for patients receiving Community Care, authorized by the Veterans Health Administration, and TRICARE
Eligible Medicare Fee for Service (FFS) patients will have $0 out-of-pocket cost for Shield. Patients with Medicare Advantage may be subject to co-pays, co-insurances, and deductibles, depending on their specific plan
CMS=Centers for Medicare and Medicaid Services.
*Medicare Advantage patients may be subject to co-pays, co-insurances, and deductibles, depending on their specific plan.
Ready to offer Shield to your patients?
Start ordering today—or connect with our team to set up your practice.

- References:
- Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology® (NCCN Guidelines®) for Colorectal Cancer Screening. V.2.2025. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed February 27, 2026. To view the most recent and complete version of the guideline, go to NCCN.org.
- Graham-Adderton C, Guerra CE, Ngo-Metzger Q, Hoang T, Raymond VM. Implementation of blood-based colorectal cancer screening: real-world adherence and outcomes. Curr Med Res Opin. 2025;41(10):1915-1920. doi:10.1080/03007995.2025.2582257
- Coronado GD, Jenkins CL, Shuster E, et al. Blood-based colorectal cancer screening in an integrated health system: a randomised trial of patient adherence. Gut. 2024;73(4):622-628. doi:10.1136/gutjnl-2023-330980
- Haynes WB Jr, Talari MP, Elmore KA, Jastan RM, Resil JJL. Blood-based colorectal cancer screening: implementation into 2 Appalachian primary care clinics. Am J Prev Med. 2025;69(1):107636. doi:10.1016/j.amepre.2025.04.006
- Data on file. Guardant Health, Inc.
- Chung DC, Gray DM II, Singh H, et al. A cell-free DNA blood-based test for colorectal cancer screening. N Engl J Med. 2024;390(11):973-983. doi:10.1056/NEJMoa2304714
- Calendar Year (CY) 2025 Medicare Physician Fee Schedule Final Rule. November 1, 2024. Accessed February 27, 2026. https://www.cms.gov/newsroom/fact-sheets/calendaryear-cy-2025-medicare-physician-fee-schedule-final-rule